|
GEM-X MO Chip, gasket attached Representative chip image
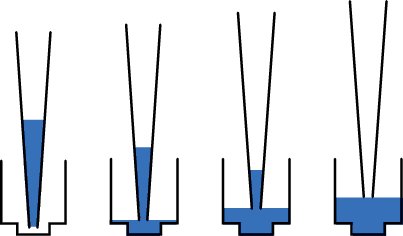
Pipette technique
|
|
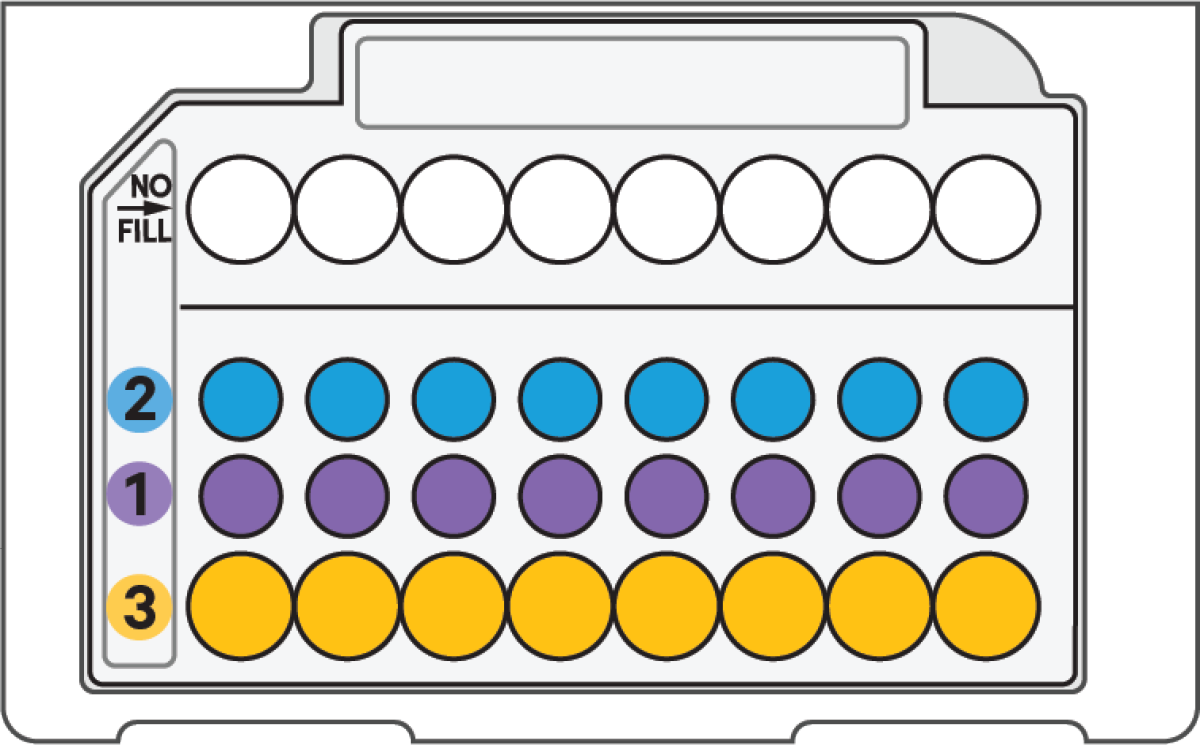
a. If loading less than 8 samples/chip, add 50% glycerol solution to each unused well in row 1, 2, and 3
DO NOT add 50% glycerol solution to the wells in top row labeled NO FILL. DO NOT use any substitute for 50% glycerol solution. |
Glycerol
|
|
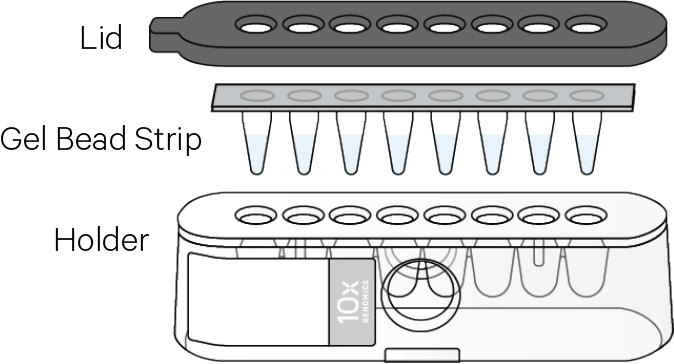
b. Prepare Gel Beads
|
Prep Gel Beads
|
|
|
|
|
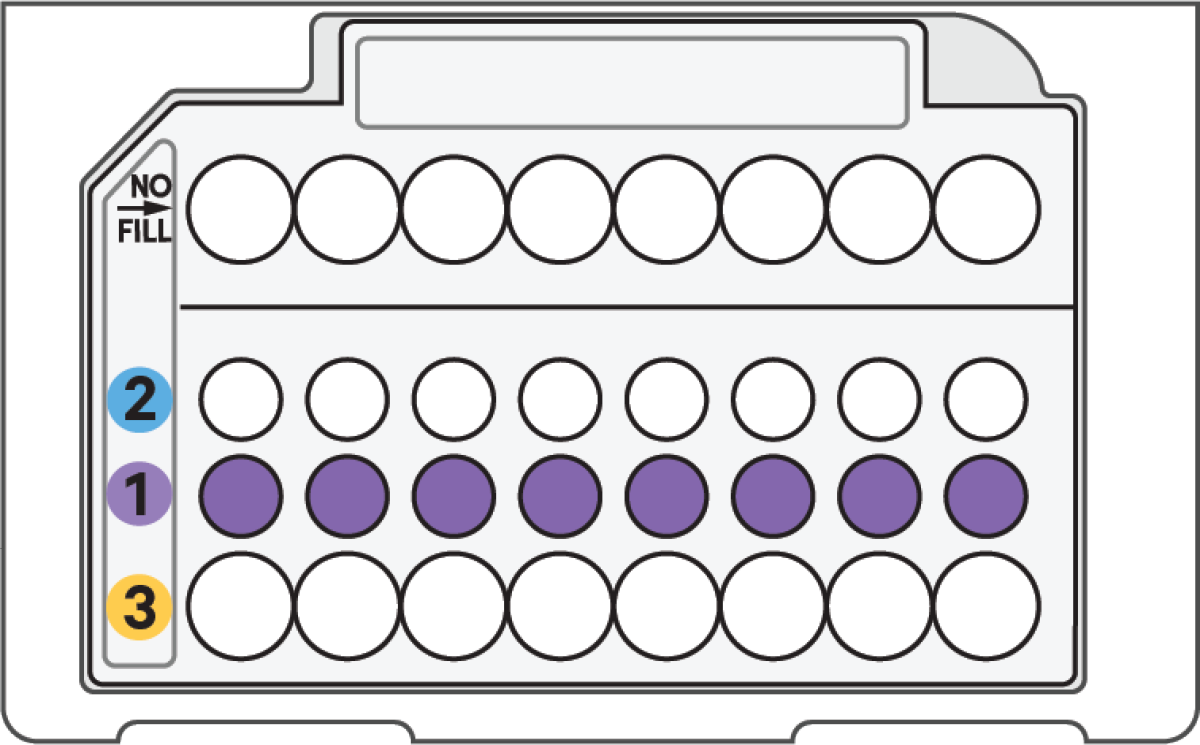
d. Load Row Labeled 1
|
60 μl Master Mix + Cell Suspension in GEM-X MO Chip
|
|
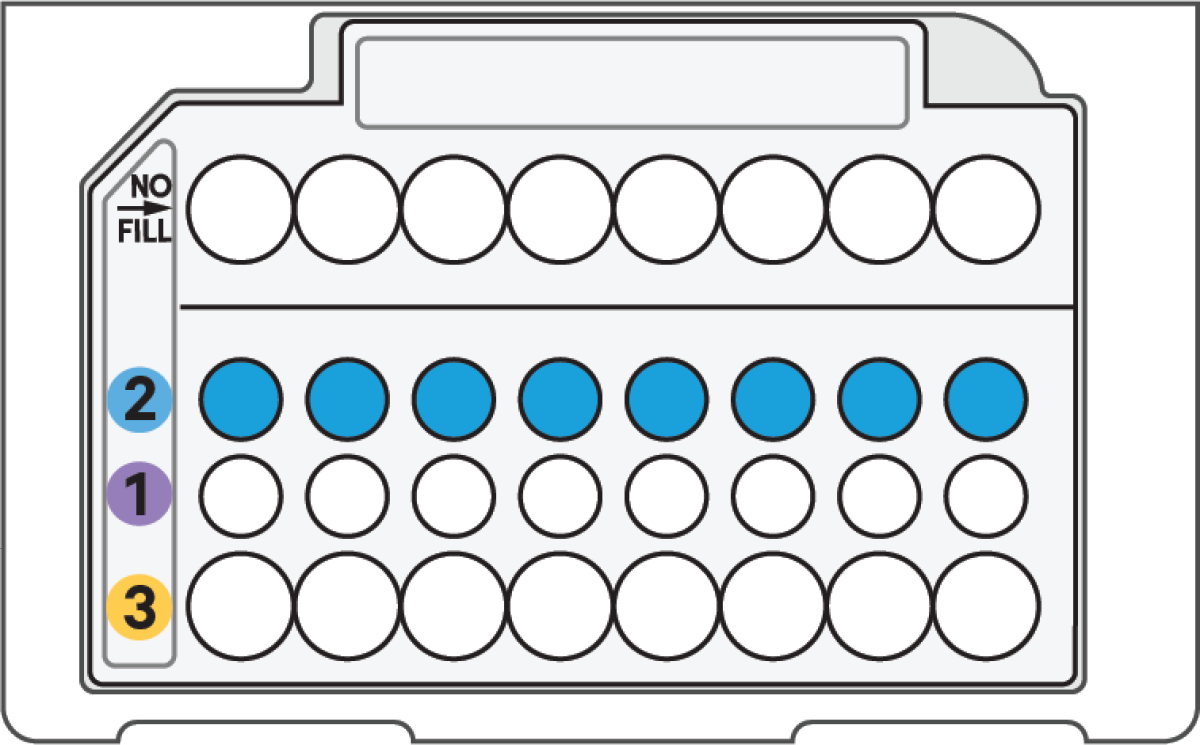
e. Load Row Labeled 2
|
60 μl Gel Beads
|
|
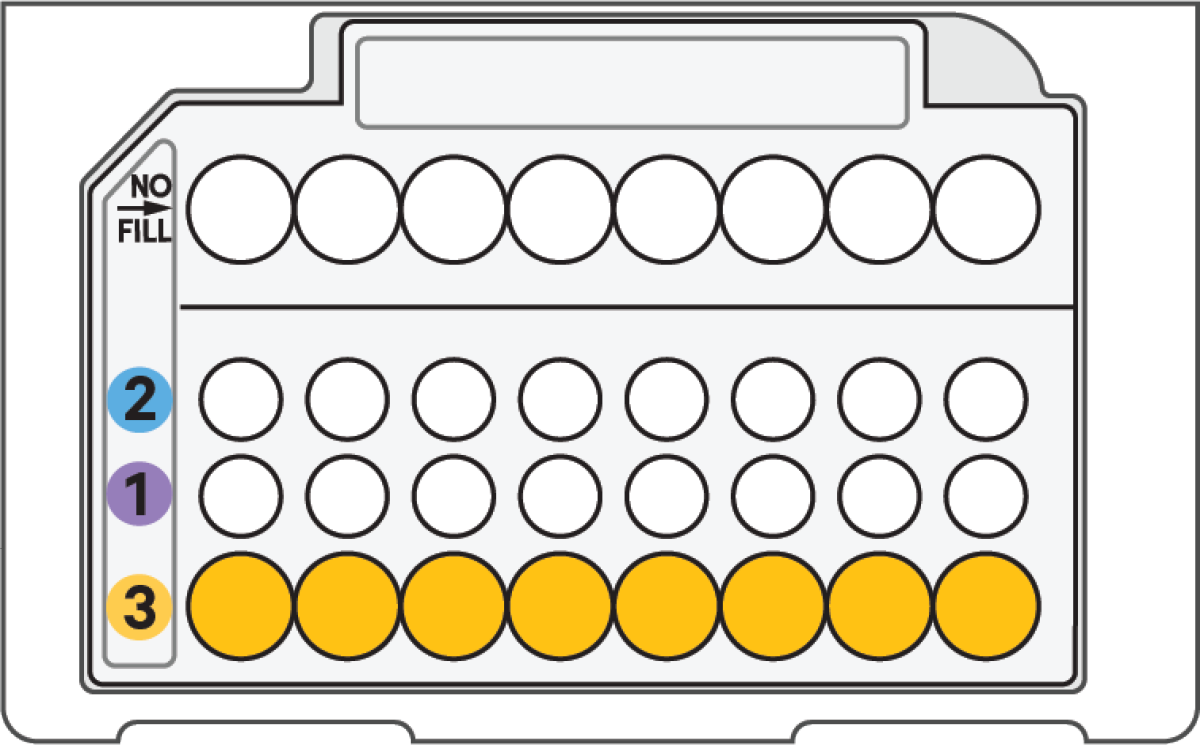
f. Load Row Labeled 3
|
250 μl Partitioning Oil B
|
|
g. Prepare for Run
Run the chip in Chromium X series instrument (X/iX) immediately after loading the Partitioning Oil B. |
GEM-X MO Chip, closed
|
*If the chip was tilted, oil spillage may appear as fluid between the chip and surface of the chip holder. It is recommended to proceed with the run in such cases. If the recovered emulsion volume appears normal, proceed with the rest of the assay. If enough oil is spilled out of the well, it can result in < 100 μl recovered emulsion volume.