4.1 Post GEM Incubation Cleanup – Dynabeads
-
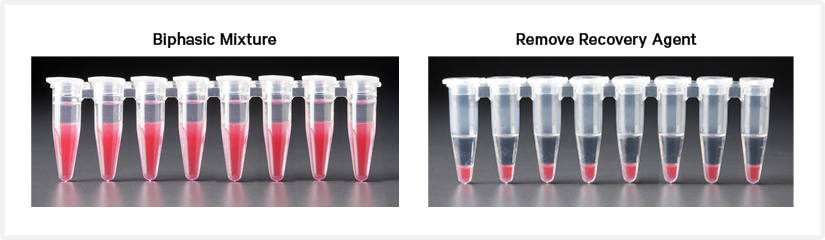
Add 125 μl Recovery Agent to each sample (Post GEM-RT) at room temperature. If using sample stored at −80°C, thaw and equilibrate to room temperature for ~15 min before adding the Recovery Agent. DO NOT pipette mix or vortex the biphasic mixture. Wait 2 min.
The resulting biphasic mixture contains Recovery Agent/Partitioning Oil (pink) and aqueous phase (clear), with no persisting emulsion (opaque).
If biphasic separation is incomplete:
-
Firmly secure the cap on the tube strip, ensuring that no liquid is trapped between the cap and the tube rim.
-
Mix by inverting the capped tube strip 5x, centrifuge briefly, and proceed to step b. DO NOT invert without firmly securing the caps.
A smaller aqueous phase volume indicates a clog during GEM generation.
-
-
Slowly remove and discard 125 μl Recovery Agent/Partitioning Oil (pink) from the bottom of the tube. DO NOT aspirate any aqueous sample.
-
Prepare Dynabeads Cleanup Mix.
Before using Dynabeads MyOne SILANE to prepare the Dynabeads Cleanup Mix:
-
 Vortex the Dynabeads thoroughly (≥30 sec) immediately before adding to the mix.
Vortex the Dynabeads thoroughly (≥30 sec) immediately before adding to the mix. -
Aspirate full liquid volume in the Dynabead tube with a pipette tip to verify that beads have not settled in the bottom of the tube. If clumps are present, pipette mix to resuspend completely. DO NOT centrifuge before adding to the mix.
2
Fixed percentage added to total to help with pipettingDynabeads Cleanup Mix
Add reagents in the order listedPN
Volume for Master Mix (μl)
Scaled Volume (μl)
Cleanup Buffer
2000088 182 Dynabeads MyOne SILANE
2000048 8 Reducing Agent B
2000087 5 Nuclease-free Water
— 5 Total
200
-
-
Vortex and add 200 μl to each tube. Pipette mix 10x (pipette set to 200 μl). Before adding ensure that there are no clumps.

-
Incubate 10 min at room temperature (keep caps open). Pipette mix again at ~5 min after start of incubation to resuspend settled beads.
-
Prepare Elution Solution I. Vortex and centrifuge briefly.
1
Elution Solution I
Add reagents in the order listedPN
Volume for Master Mix (μl)
Scaled Volume (μl)
Buffer EB
— 98
10% Tween 20
— 1 Reducing Agent B
2000087 1 Total
-
At the end of 10 min incubation, place on a 10x Magnetic Separator•High position (magnet•High) until the solution clears.
A white interface between the aqueous phase and Recovery Agent is normal.
-
Remove the supernatant.
-
Add 300 μl 80% ethanol to the pellet while on the magnet. Wait 30 sec.
-
Remove the ethanol.
-
Add 200 μl 80% ethanol to pellet. Wait 30 sec.
-
Remove the ethanol.
-
Centrifuge briefly. Place on the 10x Magnetic Separator•Low position (magnet•Low).
-
Remove remaining ethanol. DO NOT dry.
-
Remove from the magnet. Immediately add 50.5 μl Elution Solution I.
-
Pipette mix (pipette set to 40 μl) without introducing bubbles. Pipette mix 15x. If beads still appear clumpy, continue pipette mixing until fully resuspended.
-
Incubate 1 min at room temperature.
-
Place on the magnet•Low until the solution clears.
-
Transfer 50 μl sample to a new tube strip.