GEM Generation & Barcoding
|
STEP |
Normal |
Impacted |
|---|---|---|
3.2Load Chromium GEM-X Chip |
|
|
| Gasket holes are aligned with the sample and gel bead wells. |
Gasket holes are misaligned with the gel bead wells. Open and close the chip holder slowly once. One of the rows of Chromium X/iX gasket will not align with the GEM-X chip. This is normal and will not impact the assay. |
|
3.4 dAfter chip is removed from the instrument and the wells are exposed. |
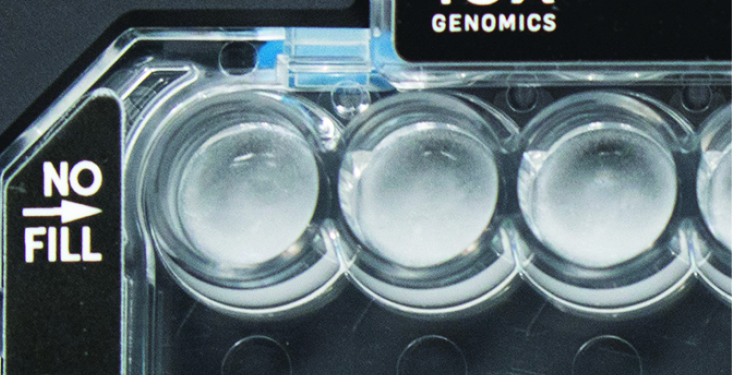
All recovery wells are similar in volume and opacity. |
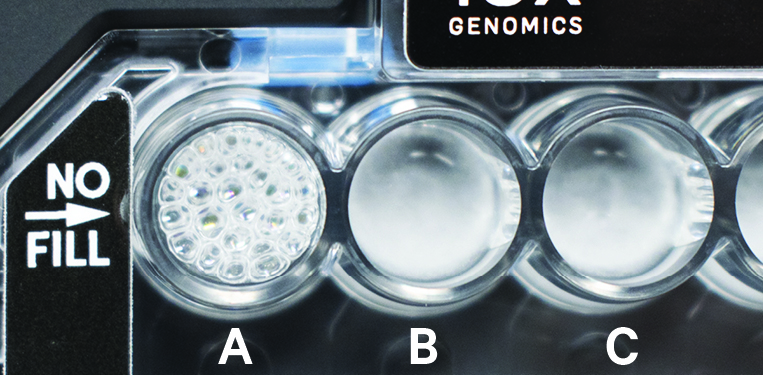
Recovery well A indicates a wetting failure. Not all wetting failures may present themselves with excess bubbles (foam). |
|
|
||
|
|
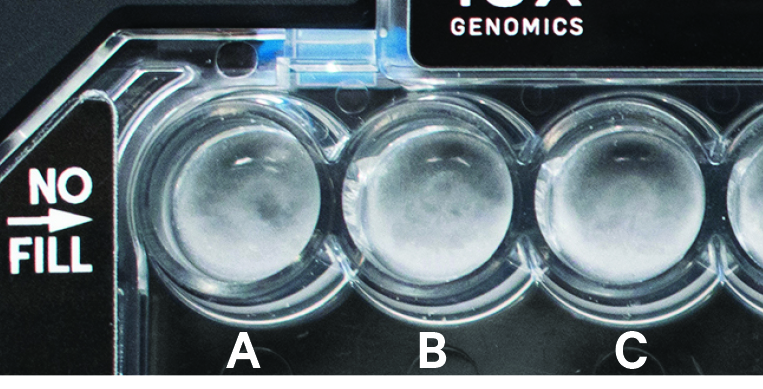
Recovery wells A-C show normal emulsions. Not all normal emulsions may have small (in size and number) bubbles in the recovery wells. Inspecting emulsions in pipette tips is recommended for diagnosing emulsion failures. |
|
3.4 eTransfer GEMs from chip |
|
|
|
All liquid levels are similar in volume and opacity without air trapped in the pipette tips. |
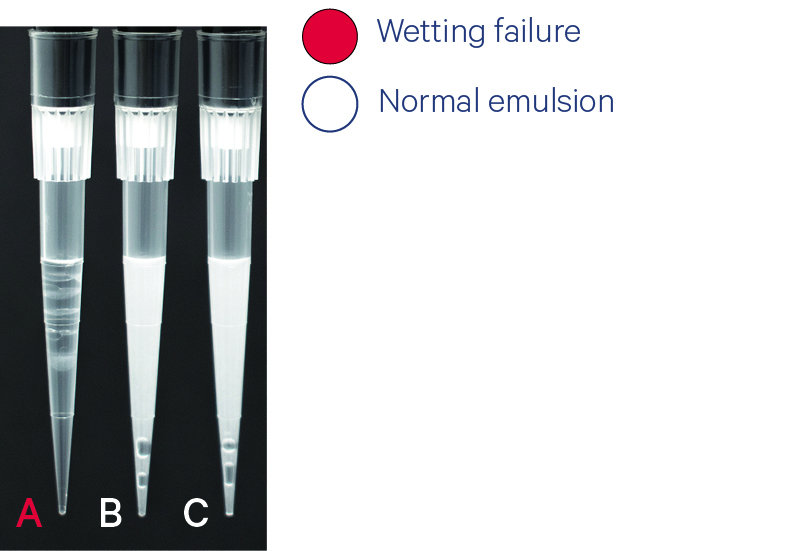
Pipette tip A shows wetting failure. Pipette tips B-C show uniform emulsions & slightly low volumes. (~95 μl) Most wetting failures will not impact emulsion volumes of other sample run on the chip. Occasionally, wetting failures may impact the emulsion volumes recovered from other lanes. This is expected and does not indicate an emulsion failure if the samples are uniform in volume and opacity (B-C). |
|
|
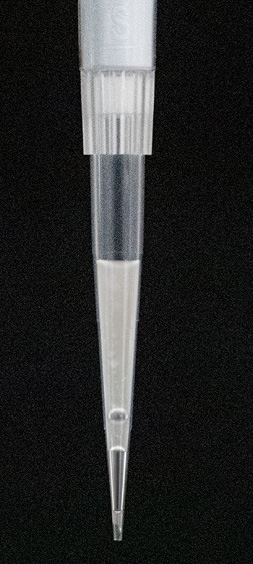
Pipette tip shows recovery of slightly lower emulsion volume (90-95 µl). This is not a clog, but a pipetting error during chip loading or emulsion recovery. It is acceptable to proceed to the next steps in this case but the cell recovery may be slightly lower. To prevent this, use a calibrated pipette and follow the loading and recovery instructions closely. |
|
|
|
|
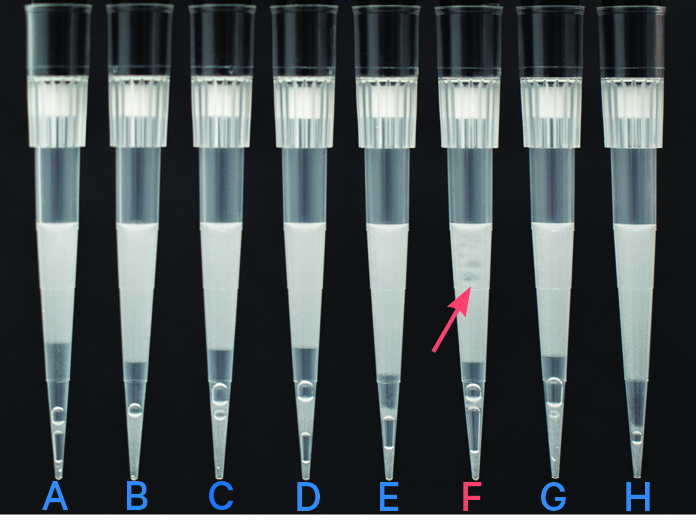
In pipette tips A-E & G-H, a portion of the emulsion displays separate layers of oil, which indicates clog. Pipette tip F indicates a clog (separate oil layers) and wetting failure (non-uniform emulsion). |
|
| Emulsion failures are not expected to be observed across the entire chip. The above images are for illustrative purposes to show a range in emulsion failures. A clog is not expected to impact other lanes on the chip. | ||
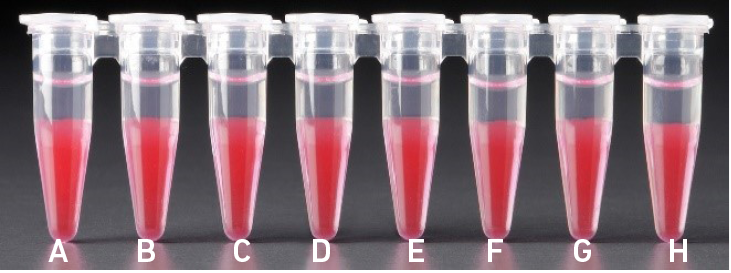
4.1 aAfter transfer of the GEMs + Recovery Agent |
|
|
| All liquid levels are similar in the aqueous sample volume (clear) and Recovery Agent/ Partitioning Oil (pink). |
Tube G indicates a reagent clog has occurred. There is a decreased volume of aqueous layer (clear). Tube C and E indicate a wetting failure has occurred. There is an abnormal volume of Recovery Agent/Partitioning Oil (pink). |
|
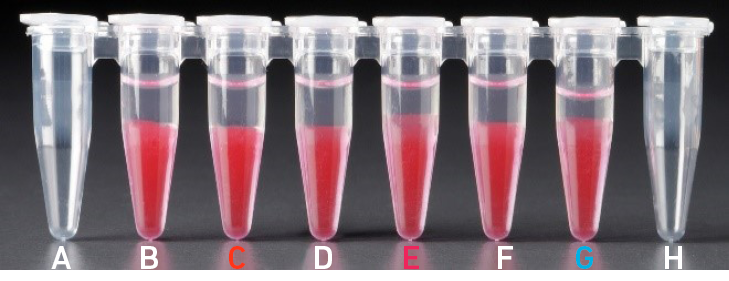
4.1 bAfter aspiration of Recovery Agent/ Partitioning Oil |
|
|
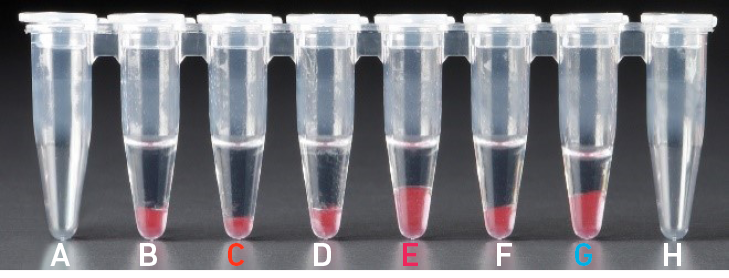
| All liquid volumes are similar in the aqueous sample volume (clear) and residual Recovery Agent/Partitioning Oil (pink). |
Tube G indicates a reagent clog has occurred. There is a decreased volume of aqueous layer (clear). There is also a greater residual volume of Recovery Agent/ Partitioning Oil (pink). Tube C and E indicate a wetting failure has occurred. There is an abnormal residual volume of Recovery Agent/Partitioning Oil (pink). |
|
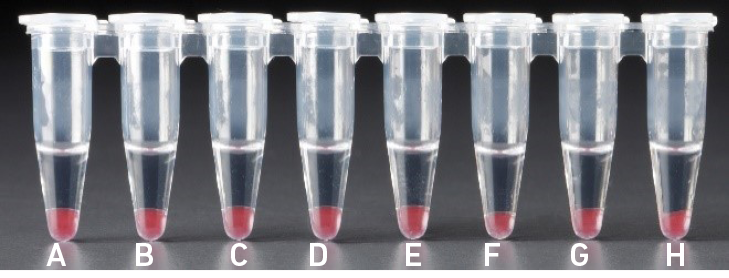
4.1 dAfter addition of Dynabeads Cleanup Mix |
|
|
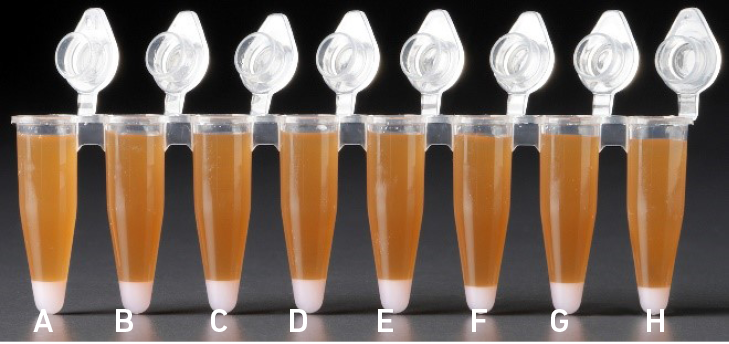
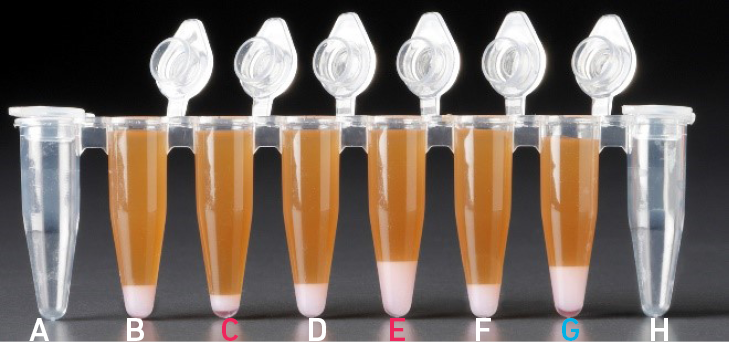
| All liquid volumes are similar after addition of the Dynabeads Cleanup Mix. |
Tube G indicates a reagent clog has occurred. There is an abnormal ratio of Dynabeads Cleanup Mix (brown) to Recovery Agent/Partitioning Oil (appears white). Tube C and E indicate a wetting failure has occurred. There is an abnormal ratio of Dynabeads Cleanup Mix (brown) to Recovery Agent/Partitioning Oil (appears white). |
|
After GEM generation, it is strongly recommended to always take a picture of:
-
Emulsion(s) in the chip (horizontal and at 45 degrees)
-
Emulsion(s) in the pipette tip/tube
-
The chip after emulsions have been recovered
If a channel clog or wetting failure is documented in one or more samples in a set:
-
Data can be derived from the remaining samples in the set by immediately proceeding with GEM-RT
OR
-
Data from all the samples in a set can be derived by immediately restarting GEM generation. If restarting GEM generation for one set of samples, ensure that a thermal cycler is available for the second set as GEM-RT incubation should start within 5 min of GEM recovery.
Contact support@10xgenomics.com for further assistance. Replacement reagents and chips may be provided for properly documented clogs or wetting failures if they are associated with runs of unexpired reagents and chips, and are reported within 30 days of the expiration date.
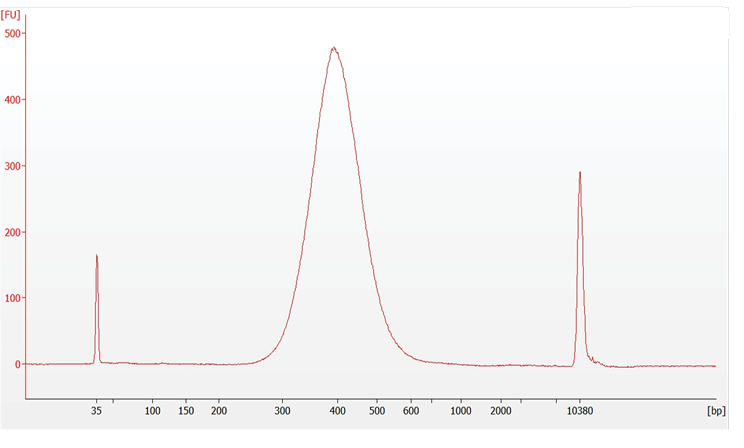
Post library Construction QC
|
Step |
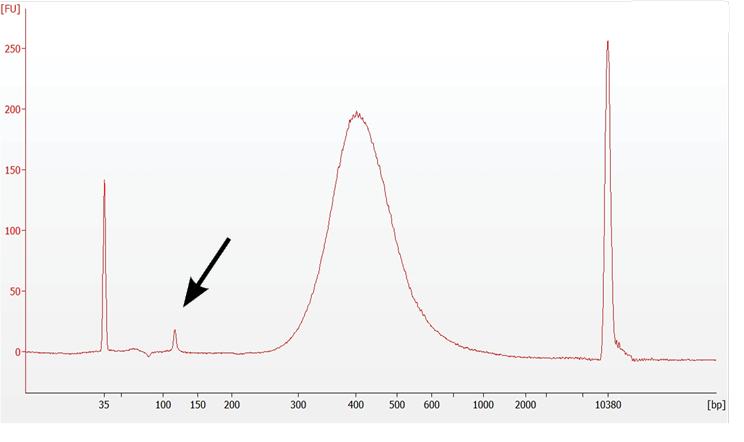
Normal |
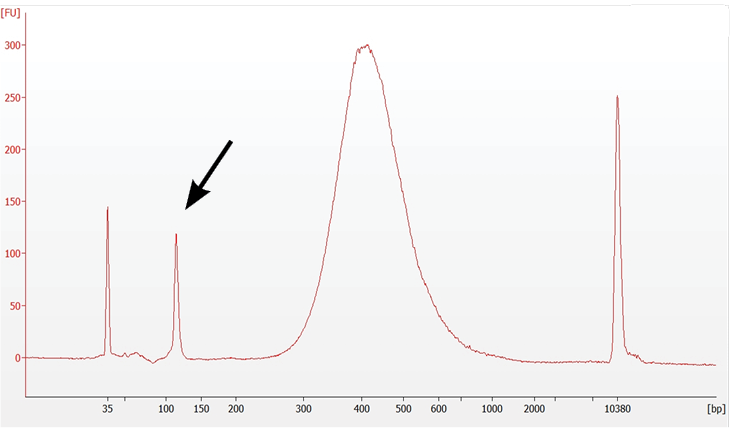
Prominent Adapter Dimer Peaks |
||||||
|---|---|---|---|---|---|---|---|---|
8.7 Post Library Construction QC (Gene Expression) |
|
|